3.1 States of matter
|
Previous
End of chapter exercises
|
Next
3.2 The kinetic molecular theory
|
Chapter 3: States of matter and the kinetic molecular theory
3.1 States of matter (ESAAK)
In this chapter we will explore the states of matter and then look at the kinetic molecular theory. Matter exists in three states: solid, liquid and gas. We will also examine how the kinetic theory of matter helps explain boiling and melting points as well as other properties of matter.
All matter is made up of particles. We can see this when we look at diffusion.
- Diffusion
-
Diffusion is the movement of particles from areas of higher concentration to areas of lower concentration.
Diffusion can be seen as a spreading out of particles resulting in an even distribution of the particles. You can see diffusion when you place a drop of food colouring in water. The colour slowly spreads out through the water. If matter were not made of particles that are constantly moving then we would only see a clump of colour when we put the food colouring in water, as there would be nothing that could move about and mix in with the water.
Food colouring in water

Diffusion is a result of the constant thermal motion of particles. In 1828 Robert Brown observed that pollen grains suspended in water moved about in a rapid, irregular motion. What is now known as Brownian motion is the random movement of microscopic particles suspended in a liquid or a gas. It is caused by collisions between these particles and the particles of the liquid or gas, and is the mechanism by which diffusion occurs.
Matter exists in one of three states, namely solid, liquid and gas. A solid has a fixed shape and volume. A liquid takes on the shape of the container that it is in. A gas completely fills the container that it is in. Matter can change between these states by either adding heat or removing heat. This is known as a change of state. As we heat an object (e.g. water) it goes from a solid to a liquid to a gas. As we cool an object it goes from a gas to a liquid to a solid. The changes of state that you should know are:
-
Boiling
- Boiling point
-
The temperature of a liquid at which its vapour pressure equals the external (atmospheric) pressure.
-
Melting
- Melting point
-
The temperature at which a solid changes its phase or state to become a liquid. The process is called melting.
-
Freezing
- Freezing point
-
The temperature at which a liquid changes its phase to become a solid. The process is called freezing.
-
Condensation is the process of going from a gas to liquid.
-
Sublimation is the process of going from a solid to a gas. The reverse process is called deposition.
-
Deposition is the process of going from a gas to a solid. The reverse process is called sublimation.
-
Evaporation is the change of a liquid to a vapour at any temperature below the boiling point. Evaporation occurs at the surface of a liquid, where molecules with the highest kinetic energy are able to escape. When this happens, the average kinetic energy of the liquid is lowered, and its temperature decreases.
If we know the melting and boiling point of a substance then we can say what state (solid, liquid or gas) it will be in at any temperature.
Figure 3.1 summarises these processes:

Figure 3.1: Changes in phase
Heating and cooling curve of water
Aim
To investigate the heating and cooling curve of water.
Apparatus
-
beakers
-
ice
-
Bunsen burner
-
thermometer
-
water

Method
-
Place some ice in a beaker.
-
Measure the temperature of the ice and record it.
-
After 1 minute measure the temperature again and record it. Repeat every minute, until at least 3 minutes after the ice has melted.
-
Plot a graph of time versus temperature for the heating of ice.
-
Heat some water in a beaker until it boils. Measure and record the temperature of the water.
-
Remove the water from the heat and measure the temperature every 1 minute, until the beaker is cool to touch.
Be careful when handling the beaker of hot water. Do not touch the beaker with your hands, you will burn yourself.
Results
-
Record your results in the following table:
Heating of ice
Cooling of boiling water
Time (min)
Temperature (in ℃)
Time (min)
Temperature (in ℃)
0
0
1
1
2
2
etc.
etc.
-
Plot a graph of time (independent variable, x-axis) against temperature (dependent variable, y-axis) for the ice melting and the boiling water cooling.
Discussion and conclusion
You should find that the temperature of the ice increases until the first drops of liquid appear and then the temperature remains the same, until all the ice is melted. You should also find that when you cool water down from boiling, the temperature remains constant for a while, then starts decreasing.
In the above experiment, you investigated the heating and cooling curves of water. We can draw heating and cooling curves for any substance. A heating curve of a substance gives the changes in temperature as we move from a solid to a liquid to a gas. A cooling curve gives the changes in temperature as we move from gas to liquid to solid. An important observation is that as a substance melts or boils, the temperature remains constant until the substance has changed state. This is because all the heat energy goes into breaking or forming the bonds between the molecules.
The following diagrams give examples of what heating and cooling curves look like:
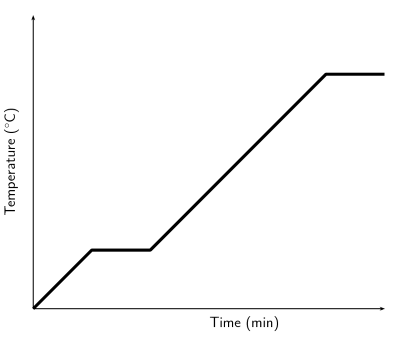
Figure 3.2: Heating curve
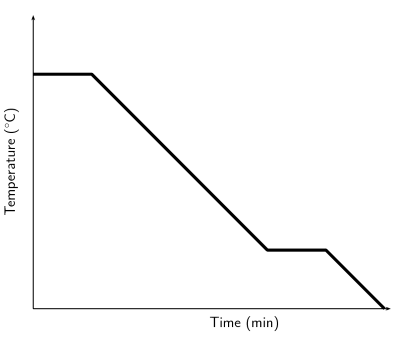
Figure 3.3: Cooling curve
|
Previous
End of chapter exercises
|
Table of Contents |
Next
3.2 The kinetic molecular theory
|
