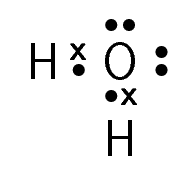
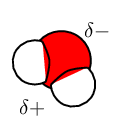
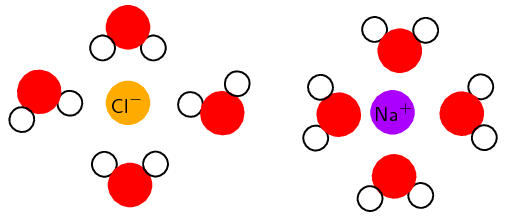
For each of the following, say whether the substance is ionic or molecular.
-
potassium nitrate (\(\text{KNO}_{3}\))
-
ethanol (\(\text{C}_{2}\text{H}_{5}\text{OH}\))
-
sucrose (a type of sugar) (\(\text{C}_{12}\text{H}_{22}\text{O}_{11}\))
-
sodium bromide (\(\text{NaBr}\))